/cloudfront-us-east-2.images.arcpublishing.com/reuters/NO2ZBBKLQRMULFDWRPMV3M2WOA.jpg)
Adele warns fans 'I'll f-ing kill you' if they throw anything at her following dangerous concert trendĪdele dared fans to throw things at her, adding she would 'f-ing kill' them if they tried, following a recent trend at concerts where attendees throw items at performers onstage. Our Standards: The Thomson Reuters Trust Principles.Failed to load news. Wait what? FDA wants 55 years to process FOIA request over vaccine data than the pandemic, the Pfizer vaccine, getting every American vaccinated," and assuring the public that the vaccine was not "'rush on behalf of the United States.'" Quoting from remarks made during the hearing before him on December 14, he wrote that “there may not be a ‘more important issue at the Food and Drug Administration. Pittman in his order made clear that the FOIA request, even if burdensome, has to be a priority for the FDA. 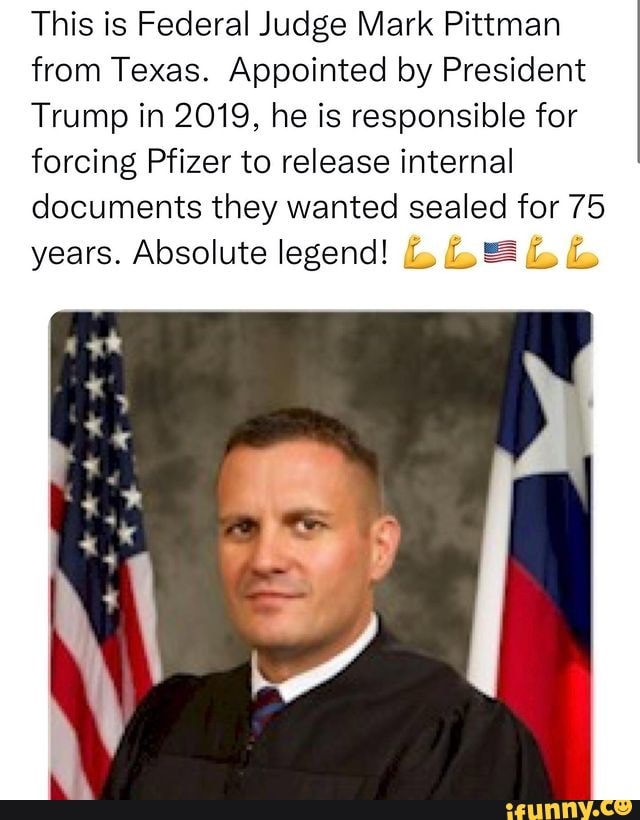
Pfizer, not a party to the suit, also did not immediately respond to a request for comment. The Justice Department, which represented the FDA in the litigation, did not immediately respond to a request for comment on Thursday evening. His clients - a group that includes more than 200 doctors, scientists, professors and public health professionals, including some who have publicly questioned the efficacy of lockdown policies, mask mandates and the vaccine itself - have pledged to publish all the information they receive from the FDA on their website. 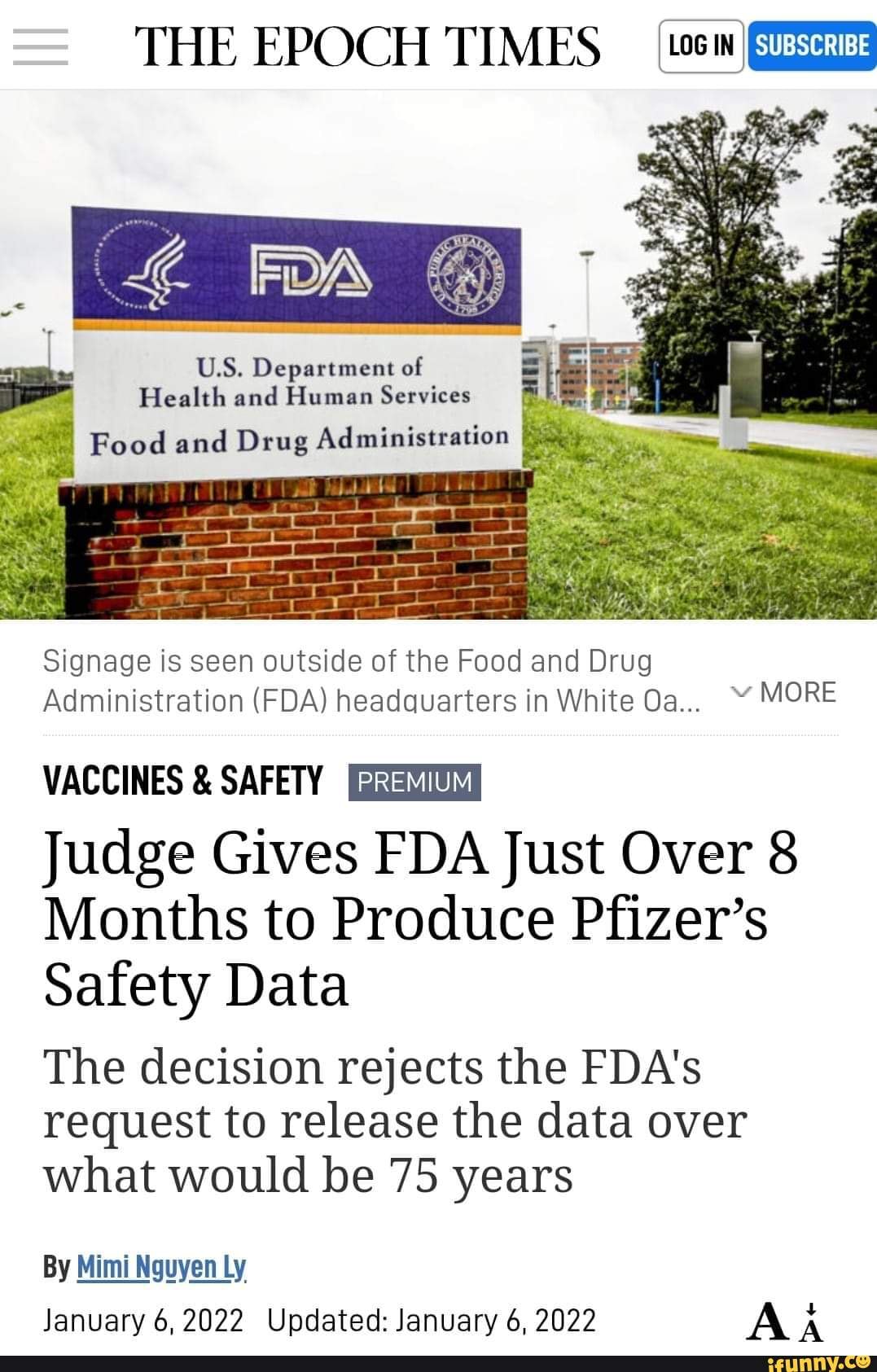
Surely some can be dispatched to pitch in at the FOIA office.Īaron Siri of Siri & Glimstad, who represents the plaintiffs, in an email said the decision "came down on the side of transparency and accountability." Burk said it takes eight minutes a page for a worker “to perform a careful line-by-line, word-by-word review of all responsive records before producing them in response to a FOIA request.”Īt that rate, the 10 employees would have to work non-stop 24 hours a day, seven days a week to produce the 55,000 pages a month (and would still fall a bit short).īut as lawyers for the plaintiffs Public Health and Medical Professionals for Transparency pointed out in court papers, the FDA as of 2020 had 18,062 employees. The office that reviews FOIA requests has just 10 employees, according to a declaration filed with the court by Suzann Burk, who heads the FDA’s Division of Disclosure and Oversight Management. Still, the FDA is likely to be hard-pressed to process 55,000 pages a month. Pittman in his order nodded to this as well, including a quote from the late senator John McCain, who said that excessive administrative secrecy “feeds conspiracy theories and reduces the public’s confidence in the government.” Making the information public as soon as possible may help assuage the concerns of vaccine skeptics and convince them the product is safe. I’ve been chronicling this fight since November and have heard from of readers who said they felt something was suspicious, even nefarious, in the FDA’s proposed slo-mo timeline. That means all the Pfizer vaccine data should be public by the end of the summer rather than, say, the year 2097.Įven if the FDA may not see it this way, I think Pittman did the agency - and the country - a big favor by expediting the document production. Rather than producing 500 pages a month - the FDA's proposed timeline - he ordered the agency to turn over 55,000 a month. While Pittman recognized “the ‘unduly burdensome’ challenges that this FOIA request may present to the FDA,” in his four-page order, he resoundingly rejected the agency’s suggested schedule. The FDA didn’t dispute it had an obligation to make the information public but argued that its short-staffed FOIA office only had the bandwidth to review and release 500 pages a month. District Judge Mark Pittman in Fort Worth, who was appointed to the bench by former President Donald Trump in 2019. The court “concludes that this FOIA request is of paramount public importance,” wrote U.S. That’s roughly 75 years and four months faster than the FDA said it could take to complete a Freedom of Information Act request by a group of doctors and scientists seeking an estimated 450,000 pages of material about the vaccine. 
A federal judge in Texas on Thursday ordered the Food and Drug Administration to make public the data it relied on to license Pfizer’s COVID-19 vaccine, imposing a dramatically accelerated schedule that should result in the release of all information within about eight months.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
